Utilization of stable isotopes such as deuterium (2Hydrogen), 18Oxygen, 13Carbon and 15Nitrogen is commonplace in a wide range of fields spanning from environmental analysis to clinical diagnosis and research to pharmacology. They constitute a power tracer tool commonly deployed to answer specific research questions.
Assessment of drug pharmacology
Pharamacokinetics
Mechanism of action
Mechanism of troxicity and adverse effects
Assessment of drug products or drug delivery systems
Bioavailability
Release profile of drug delivery systems
Assessment of patients
Phenotyping
Monitoring drug treatment effect
Clinical toxicology
| Element | Atomic Number | Parent Atom | Stable Isotope | Abundance (In Nature) |
|---|---|---|---|---|
Hydrogen | 1 | 1H | 2H | 0.015% |
Nitrogen | 6 | 12C | 13C | 1.1% |
Carbon | 7 | 14N | 15N | 0.4% |
Oxygen | 8 | 16O | 17O | 0.04% |
Paradigm Shift in Drug Discovery
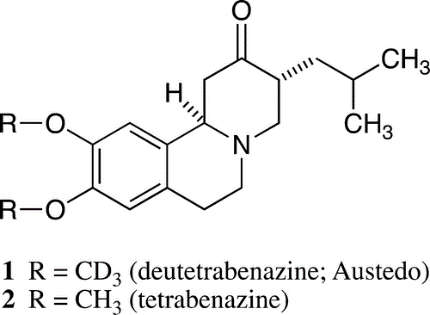
Recent discovery of the utility of the deuterated drug products have generated excitement and investment in the field. An excellent case study is Deutetrabenazine. The fundamental principle is to prolong the residence time of the active drug species in plasma to achieve greater efficacy and/or to avoid adverse side effects.
Deutetrabenazine is ananalog of the old drug tetrabenazine, with the two methoxy groups in the latter being replaced by a pair of trideuteromethoxy groups, thereby altering the rate of metabolism to afford greater tolerability and an improved dosing regimen.
Deuterium substitution impedes oxidative metabolism of the methoxy groups, in an excellent demonstration of the primary kinetic isotope effect (KIE). This type of KIE has often been used to probe chemical reaction mechanisms, but it rises to a practical level by substantially improving drug entity 1 over 2. In fact, the reaction rate of a C−D bond can be 10 times slower, or even more, than the rate for the corresponding C−H bond.
Development of a deuterated analogue of an approved drug may be derisked and expedited via a 505(b)(2) regulatory pathway. By using this approach, an applicant relies on studies completed for approval of the parent nondeuterated drug, even though the studies for it “were not conducted by or for the applicant and for which the applicant has not obtained a right of reference or use from the person by or for whom the investigations were conducted”, according to FDA document 21 U.S.C. 355(b)(2).
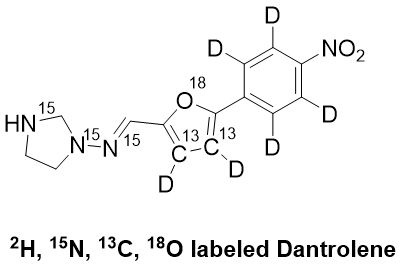
AAPharmaSyn has devoted significant resources to building competencies in bringing stable isotope labeled compounds to the market. Our expertise includes introducing a stable isotope with a complex molecule as well as making stable isotope labeled compounds such as 18O labeled potassium carbonate.
We carry an extensive inventory of stable isotope labeled compounds and are able to expeditiously prepare new compounds:
Please write to us if you have a consistent need for any stable isotope labeled compounds. We offer industry-leading pricing for long-term supply agreements as well as volume discounts.
